Revised Second Edition | Available Now
All sterile compounding procedures in this courseware are aligned with
USP <797> (USP-NF 2023, Issue 1) and USP <800> (USP43-NF38).

About the Courseware
Authors:
This courseware provides instruction and training for the compounding of sterile parenteral preparations, a specialized, advanced practice of pharmacy technicians in hospitals, long-term care facilities, and home health care. This practice requires mastery of aseptic technique, or the procedures that avoid the introduction of pathogens into the sterile compounding environment and the preparations themselves.
Introducing the history, concepts, procedures, and revised USP <797>, this courseware leads students through a step-by-step exploratory or procedural lab that provides an opportunity for students to demonstrate their understanding of these major concepts.
Unit 1: Exploring the Basics of Sterile Compounding
Chapter 1: Introduction to Sterile Compounding
Chapter 2: Medication Safety
Chapter 3: Law, Ethics, and Quality Assurance
Chapter 4: The Sterile Compounding Environment
Chapter 5: Sterile Compounding Supplies
Chapter 6: Medication Orders and Labeling
Chapter 7: Calculations for Sterile Compounding
Unit 2: Compounding Sterile Preparations
Chapter 8: Hand Hygiene and Garbing
Chapter 9: Cleaning the Horizontal Laminar Airflow Workbench
Chapter 10: Large-Volume Parenteral Preparations
Chapter 11: Small-Volume Parenteral Preparations
Chapter 12: Ampule-Based Preparations
Chapter 13: Narcotic Analgesic Preparations
Chapter 14: Pediatric Preparations
Chapter 15: Parenteral Nutrition Preparations
Chapter 16: Hazardous Drug Preparation
Appendices
Appendix A: Useful Reference Tables
Appendix B: Procedural Lab Competency Assessment Checklists
Appendix C: ASHP/ACPE Accreditation Standards
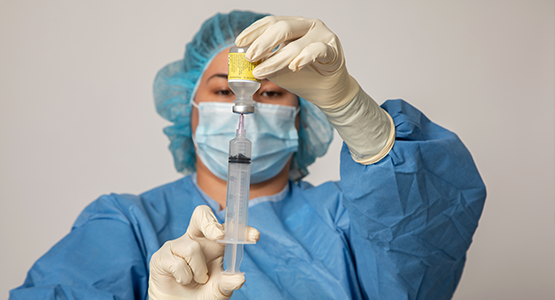
Key Features
- Aligns with revised USP <797> (USP-NF 2023, Issue 1) and USP <800> (USP43-NF38)
- Instructs and trains students in sterile compounding and aseptic technique and its real-world applications in a sterile compounding facility
- Offers brief videos that demonstrate the nine procedural labs
- Includes updated videos and photography
- Offers supplemental reference materials in the appendices
- Integrates with LTI-supported learning management systems, including Canvas, Blackboard, D2L, and Moodle
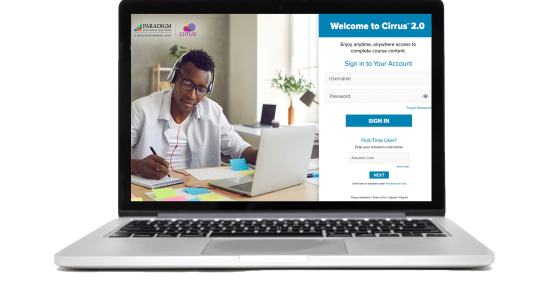
Cirrus™ 2.0 for Sterile Compounding and Aseptic Technique
Sterile Compounding and Aseptic Technique, powered by the Cirrus platform, integrates seamlessly with Blackboard, Canvas, D2L, and Moodle.
To strengthen student understanding, Cirrus provides interactive, independent learning experiences, including:
- self-assessment opportunities
- critical-thinking, application, and research activities
- quizzes and exams
- procedural lab videos
Course content is delivered in a series of scheduled assignments that report to a grade book, thus tracking student progress and achievement.
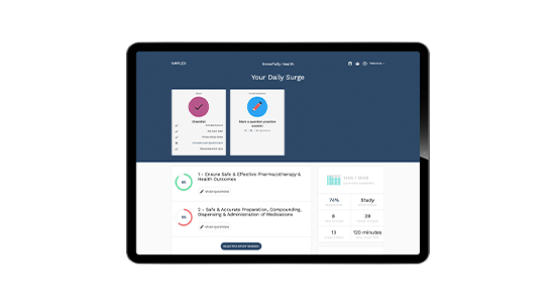
Access to PTCE Exam Review from PharmCon freeCE
Included with all of Paradigm’s Pharmacy Technician titles, this tool generates personalized study plans based on each student’s individual strengths and knowledge gaps. The PTCE Exam Review from PharmCon freeCE helps students identify the fastest path to a passing score and even indicates when they are ready to pass—taking the guesswork out of PTCE study.
PTCE Exam Review from PharmCon freeCE features:
- Optimized PTCE-aligned content
- Award-winning adaptive learning technology
- ReadySCORE proprietary exam readiness indicator
- 700+ multiple-choice questions
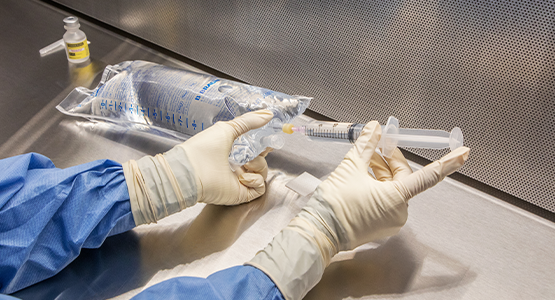
Instructor Resources in Cirrus
Utilize our free instructor resources that include:
- course-planning guidelines
- syllabus models
- teaching hints and activities
- quizzes and exams
- answer keys
- instructor rubrics
- handouts
- and more!

USP <797> Changes Incorporated in Second Edition
On November 1, 2022, the USP published a revision of USP General Chapter <797> Pharmaceutical Compounding—Sterile Preparations. In light of that, our second edition of Sterile Compounding and Aseptic Technique: Concepts, Training, and Assessment for Pharmacy Personnel aligns with this current, accepted USP <797> standard and, therefore, provides the necessary material to educate and train students in this specialized area of pharmacy practice. The revised USP <797> goes into effect November 1, 2023.

Competency Guide
See how our courseware aligns to the entry-level and advanced-level key elements of the revised ASHP/ACPE Accreditation Standards for Pharmacy Technicians Education and Training Programs.
Request Free Brochure
A Complete Solution
Our Pharmacy Technician series is a complete program solution that develops the skills and knowledge students need to become certified pharmacy technicians. Our full suite of products, competency mapping guides, and program support are designed for success and meet ASHP and ACPE standards.
Browse Courseware
